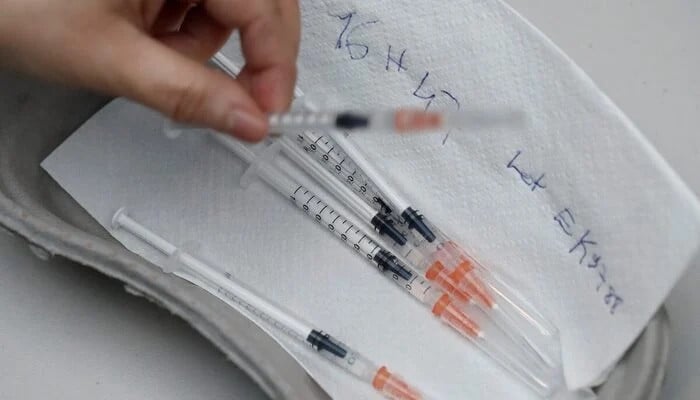
- Samples show so-called single-use syringes can be reused.
- Regulatory body says it is aware of compliance gaps.
- Violation found in Peshawar, Multan, Jacobabad, other regions.
ISLAMABAD: Reusable syringes, including some falsely marketed as auto-disable devices, continue to be manufactured, supplied and used across Pakistan despite a nationwide ban, raising concerns over enforcement failures and risks of blood-borne infections, an investigation by The News uncovered.
Even more troubling, syringes are being fraudulently marketed as “auto-disable” while in reality remaining fully reusable, according to the report.
This deceptive practice is undermining a crucial infection control measure designed to prevent the reuse of syringes and curb the spread of disease.
Samples collected from multiple cities show that syringes marketed and sold as single-use safety devices can be easily reused, with plungers moving freely back and forth instead of locking after one use, raising serious concerns about regulatory failure and the unchecked spread of blood-borne infections.
Drug Regulatory Authority of Pakistan, however, said they are aware of compliance gaps and are taking steps to enforce their directives. Officials of the provincial authorities also claim that they are actively ensuring compliance with the ban, rejecting the impression of any laxity on their part.
Field findings indicate that such syringes are available in Peshawar, Multan, and Jacobabad, with reports of their presence in rural as well as some Urban areas across the country, suggesting that the ban on 2ml, 3ml and 5ml conventional disposable syringes largely exists on paper.
Investigations by this scribe further traced a significant number of these syringes to manufacturing units in the Gadoon Amazai industrial zone, where factories are allegedly producing and distributing reusable syringes across the country, bypassing regulatory scrutiny. Healthcare workers on the ground say the consequences are already visible.
“These syringes are easily available in markets and pharmacies, and despite repeated complaints, no action is taken,” a healthcare worker at the Jacobabad Institute of Medical Sciences said, requesting anonymity.
“Quacks charge Rs50 to Rs100 per injection and reuse the same syringe on multiple patients to cut costs.”
Jacobabad, already a hotspot for hepatitis C, is now witnessing a growing number of HIV cases, with unsafe injection practices cited as a key driver.
“The ban on conventional syringes was a genuine policy victory, but enforcement has been virtually absent,” said Syed Omer Ahmed, Chairman of the Healthcare Devices Association of Pakistan. He warned that the problem is deeply rooted in the healthcare delivery ecosystem.
“In the case of medicines, wholesale markets were often blamed for spurious products, but for medical devices it is the quack practitioner network that sustains this system,” he said.
Citing a letter issued by the DRAP on April 22, 2026, the authority’s officials said a nationwide market survey has been ordered to assess compliance with the ban and identify violations, warning that strict legal action would be taken against non-compliant products.
The document reiterates that the import and manufacture of conventional disposable syringes have been banned since July 31, 2021, and that all registrations for such products were cancelled, while directing provincial drug authorities and inspection teams to verify whether banned syringes are still being sold in the market.